All reusable dental instruments need to be cleaned and sterilised to prevent cross infection during dental procedures: an alternative to this time-consuming process is MDDI’s global range of pre-wrapped sterile single-use instruments.
These instruments are an ideal solution for ensuring compliance in infection control and CQC protocols.
MDDI has been providing single-use solutions to the dental industry globally for more than 15 years and the company partners with the world’s leading distributors.
Due to the instruments being sterilised at manufacture, they are sterile at the point of use, easy to open dispensers and packaging increases the efficiency of oral examinations and restoration procedures.
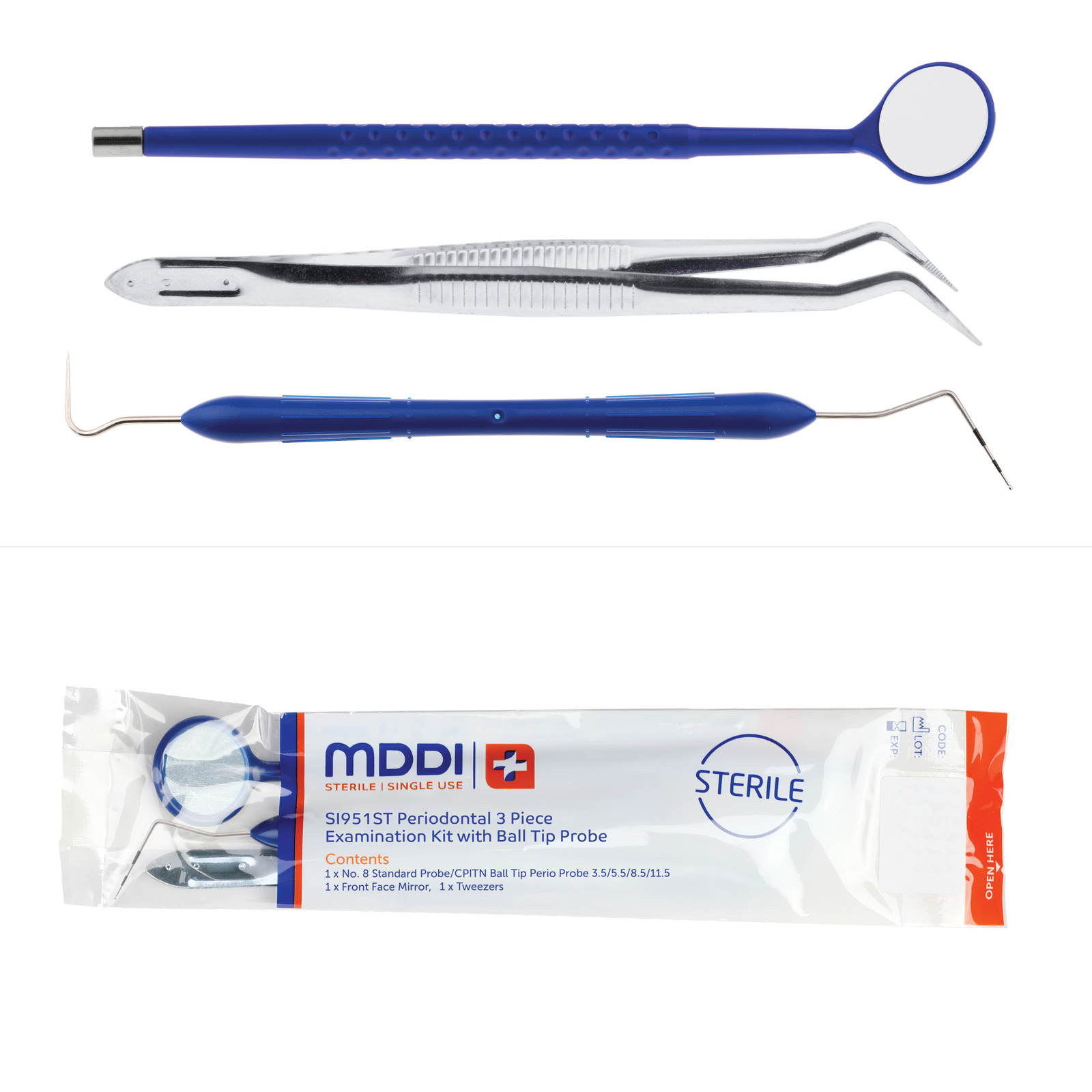
The range includes the very popular three piece exam kit, SI951ST, containing a front face mirror, examination tweezers and a double-ended probe with CPITN (WHO) tip.
This kit, retailing at £115 for 100 kits, is just one example of MDDI’s extensive range.
All kits are supplied with two and half years’ expiry from manufacture and from October 2018 GS1 data matrices have been included on all levels of packaging.
For more details, call 01535 657 209 or email the team at [email protected].


